![]()
Zhengzhou Central Hospital of Traditional Chinese Medicine,Zhengzhou Henan 450000,China
【Abstract】Asbreastcancer relative survival continues to increase, many breast cancer patients face many issues, includingrecurrenceof cancer and cancer-related side effects that impact several aspects of their quality oflife.With breast cancer patients living longer, there is more of a concern for negative breast cancer outcomes. Althoughphysicalactivityisanaffordableandrelatively convenient way to improve breast cancer outcomes, onlyaboutone-thirdofbreast cancer survivors engage in the recommended level of physical activity. This article reviewsarticlespublishedtodatetoexaminewhetherhome-based physical activity interventions are effective inimprovingphysicalactivityandother outcomes among breast cancer survivors who have completed primary therapyforthedisease. Thepresentreviewisbased upon bibliographic searches in PubMed and CINAHL andrelevantsearchterms. ArticlespublishedinEnglishfrom1980 through February 28, 2019 were identified. Atotalof 360 article citations were identified in PubMed and non-duplicates in CINAHL. After screening the abstractsorfulltextsof these articles and reviewing the references of previous review articles, 20 studies that met theeligibilitycriteria. Threeofthestudieswerepre-/post-testtrialsand 17were randomized controlled trials. Home-basedexerciseprogramsareeffectiveinimprovingphysical activity among breast cancer survivors who havecompletedprimarytherapyforthedisease. Home-based exercise programs such as walking programs offer aconvenientandaffordableoptionforwomenwhowish to increase their physical activity and maintain a healthy lifestyle.
【Keywords】breast cancer survivors, physical activity, women
1 Introduction
The five-yearrelativebreastcancer survival rate in theUScontinues to increase and is now about 91%[1] .Astherate increases, many breast cancer patients face
manyissues, includingrecurrenceof cancer and cancer- related side effects that impact several aspects of their qualityoflife[2] . To reduce risk of cancer recurrence,theAmericanInstitutefor Cancer Research also recommendsthatcancer survivors meet physical activity guide- lines (AICR)[3]. Of women diagnosed with breast can- cer, 50–96% experience weight gain during treatment[4] . Thisweightgainafterdiagnosisusually ranges between2.5 and 6.2 kg(5.5 to 13.6 lbs)[5] . Among breast cancersurvivors (BCSs),physical activity improves physi- calfunctioning, cardiovascular fitness, emotionalwell- being, and psychological adjustment, while lowering fa- tigue, depression, and anxiety, and helping to maintain a healthybodyweight[6] . Inaddition, studiessuggest thattheimmunologicalstatus of breast cancer patients improvesafterphysicalactivity[7]. However, levels of physicalactivityinthispopulationarelow.
Physical inactivity and excessive weight gain that canoccurfollowingbreastcancertreatment increases theriskofbreastcancer recurrence, other chronic dis- eases, andall-causeandbreast cancer-related mortal- ity[8] . Physical inactivity increases the risk of obesity, whichincreasescirculatingestrogenlevelsand mortal- ity.Exercise can lower circulating levels of estrogen and potentiallyreducetumorproliferation. Although physi- calactivity is an affordable and relatively convenient way toimprovebreastcancer outcomes, only about one-third ofbreastcancersurvivorsengageinthe recommended levelof physical activity[9] .
Home-basedexerciseprograms, including walking programs, offer a convenient and affordable option for womenwhowishtoincrease their physical activity and maintainahealthylifestyle. This manuscript reviews articlespublishedtodatetoexamine whether home-based physicalactivity interventions are effective in increasing physical activity and improving other outcomes among breastcancersurvivorswhohavecompletedprimary therapy for the disease (adjuvant chemotherapy, radia-
tion, orsurgery).
2 Methods
The present review is based upon bibliographic searches in PubMed and CINAHL (CummulativeIn- dextoNursingandAllied Health Literature) and rele- vant search terms. Articles published in English from 1980 through February 28, 2019 were identified using thefollowingMeSH (MedicalSubjectHeading) search termsandBooleanalgebra commands: home based AND physical activity AND breast cancer. The follow- ing MeSH search terms and Boolean algebra commands were also used: walking intervention AND breast can- cer. Thesearcheswerenot limited to words appearing in thetitleofanarticlenorto studies in a particular country orgeographicregionofthe world. The references of re- viewarticleswerealso reviewed (Bluethman etal. 2015; Paxtonetal. 2019). Informationobtained from biblio- graphicsearches (titleand topic of article, information inabstract, studydesign, and key words) was used to de- termine whether or not to retain each identified article. OnlystudieswritteninEnglish that examined the impact of breast cancer survivorship care plans on health out- comeswereeligible for inclusion.
3 Results
Atotalof 360 article citations were identified inPubMed and non-duplicates in CINAHL (Figure 1). Af-ter screening the abstracts or full texts ofthesearticlesandreviewingthereferencesofprevious review articles,wewereleftwith 20 studiesthat met the eligibility crite-ria. Three of the studies were pre-/post-test trials, and 17were randomizedcontrolledtrials.
Pintoetal.[10] conducteda 12-weekrandomizedcon- trolled trial of physical activity counseling deliveredvia telephone, combined with weekly exercise tip sheets (Ta- ble1). Eighty-sixwomenwhohadcompletedther- apyforstage 0-IIbreastcancerwereenrolledinthe trial. The physical activity group reported significantly moretotalminutesof physical activity and more minutes of moderate-intensityphysicalactivitythanthecontrol group (p= 0.001).
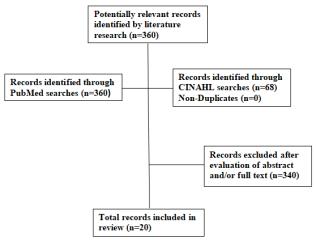
Figure1.Flowchartof record selection process
Inaneight-weekpre/post-testtrial of a community
intervention that combined the use of pedometers with scheduling,goal setting, and self-assessment, Wilson et al.[11] found that the intervention led to significant in-creasesinstepswalkedper day and improved attitude towardexercise, aswell as significant decreases in body mass index and other anthropometric measures. Twenty-fourAfricanAmericanbreast cancer survivors were enrolledinthetrial.
Vallance etal.[12] conducted a 12-week randomized controlled trial with four arms: i) standard public healthrecommendation for physical activity; ii) breastcancer- specificphysicalactivityprintmaterials; iii) useofa step pedometer; or iv) a combination of print materials anduseofasteppedometer. Physicalactivityincreased by threeminutes/weekinthestandardrecommendation groupcomparedwith 70 minutes/weekin the print mate- rialgroup (p = 0. 117), 89 minutes/week in the pedome- tergroup (p = 0.017), and 87 minutes/week in the com- binedgroup (p = 0.022). For brisk walking, all three in- terventiongroupsreportedsignificantly greater increases thanthe standard recommendation group. The combined groupalsoreportedsignificantlyimprovedquality-of- life(p = 0.003) and reduced fatigue (p = 0.052).
In a 12-week randomizedcontrolledtrialofahome- basedwalkingintervention, Matthewsetal.[13] found that intervention participants reportedasignificantly greaterincreaseinwalkingforexercisethanthecontrols (p= 0.01). In a 12-week randomized controlled trial of two home-based exercise programs (aerobic exercise, re- sistancetraining), YuenandSword[14] found a significant reductionin fatigue among participants in the aerobic ex- ercise group compared with the resistance exercise group (p = 0.006). In addition, there was a significant improve- mentinthedistancewalkedduringa 6-minute walk test intheresistanceexercisegroup (p = 0.009).
Payneetal.[15] conducted a randomized controlled trialof a home-based walking program among 20 womenreceivinghormonaltherapy for breast cancer. There wasasignificant improvement in sleep scores in the inter-
vention group comparedwiththecontrolgroup (p = 0.007). Serotonin levels were also significantly differentbetweengroups (p= 0.009).
Table1.Studiesofhome-basedphysical activity interventions for breast cancer survivors
activitycounselingdelivered via telephone, combinedwithweeklyexercisetipsheets | ||||
Wilsonetal., 2005 | 8-weekpre-/ post-test trial of a community interventionusingpedometerswith scheduling, goalsetting, andself-assessment | Stepswalkedperday, body massindex, and attitudes | 24 AfricanAmericanbreast cancersurvivors | Significantincreasesinstepswalkedperday and attitude toward exercise werereported, as well as significant decreases in body mass index and other anthropometricmeasures |
Vallanceetal., 2007 | 12-weekrandomizedcontrolledtrialwith 4 arms: standardpublichealthrecommendationfor physicalactivity; breastcancer-specific physical activity print materials; a step pedometer; or a combinationofprintmaterials and step pedometers | Self-reported moderate/vigorousphysical activityminutesperweek. Secondaryoutcomeswere quality-of-life, fatigue, self- reportedbrisk walking, and objectivestepcounts | 377 women who had completedtherapyforstageI- IIIabreastcancer | Physicalactivityincreased by 3- minutes/week in the standard recommendationgroupcomparedwith70 minutes/week in the print materialgroup (p=0. 117, 89 minutes/weekinthepedometergroup (p=0.017), and 87 minutes/weekinthe combined group (p=0.022). For briskwalking, allthree intervention groups reported significantly greater increasesthanthestandardrecommendationgroup. The combined group alsoreportedsignificantlyimprovedquality-of-life (p=0.003) and reduced fatigue(p=0.052). |
Matthewsetal., 2007 | 12-weekrandomizedcontrolledtrialofa home- basedwalking intervention | Self-reportedphysicalactivity | 36 breast cancer survivors | Intervention participants reported a significantly greater increase in walking forexercise (p=0.01) |
Yuen& Sword, 2007 | 12-weekrandomizedcontrolledtrialoftwohome- basedexerciseprograms(aerobic exercise, resistancetraining) | Fatigue, functional capacity (6-minutewalktest) | Therewas a significant reduction in fatigue among participants in the aerobic exercise group (p=0.006). There was a significant improvement in thedistanceof the 6-minute walk test in the resistance exercise group (p=0.009) | |
Fatigue, sleep disturbances, | ||||
Payneet al., 2008 | Randomizedcontroltrial of a home-based walkingprogram | depressivesymptoms, and biomarkers (cortisol, serotonin,interleukin-6, | 20 women receiving hormonal therapyfor breast cancer | Therewasasignificant improvement in sleep scores in the intervention groupcomparedwiththecontrolgroup (p=0.007). Serotonin levels were alsosignificantly different between groups (p=0.009) |
bilirubin) | ||||
Yangetal., 2010 | 12-weekrandomizedcontrolledtrialofahome- based walking program | Symptom severityscoreand mooddisturbance | 40 breast cancer patients (stageI-IIIa) receivingan aromataseinhibitor | Womenintheexercisegroupreportedsignificantlylower symptom severity scores (p<0.01) andmooddisturbance(p=0.02) compared with those in the controlgroup |
Kimet al. 2011 | 12-weekrandomizedcontrolledtrialofahome- basedstage-matched exercise and diet intervention (telephonecounselinganda workbook | Stage ofmotivational readinessforexercise and diet, physical activity, diet quality, quality-of-life, fatigue, anxiety, depression | 45 breast cancer survivors, stage 0-III | Theinterventiongroupshowedsignificantly greater improvement in motivationalreadinessforexercise (p<0.006) and diet (p<0.001), emotional functioning (p=004), fatigue (p=0.001), and depression (p=0.035) |
Hatchettetal., 2013 | 12-weekrandomizedcontrolledtrialofanemail physicalactivityintervention | Self-reportedphysicalactivity | 74 breast cancer survivors | Significantdifferencesinlevelsofphysicalactivitywereobservedbetween groupsat 6 (p=0.001) and 12 weeks (p<0.001) |
Spectoret al., 2014 | Pre-/post-testtrial of a home-based aerobic and resistance training exercise intervention (motivationalinterviewing and weekly telephone calls) | Self-reportedandobjectively assessedphysicalactivity, quality-of-life, andfatigue | 17 African Americanwomen whohadcompleted therapy forstage 0-IIIa breast cancer, whowerecurrently sedentary | Therewasasignificantincreaseintotalminutesofweeklyphysicalactivity (p=0.001). Total quality-of-life and fatigue scores improved, but neither wassignificant. |
Denysschenetal., 2014 | 8-weekpre-/ post-test trial of a home-based exerciseprogram (resistanceexercisesandself- selectedaerobicexercise) | Anthropometry and functional performanceand cardiovascularendurance(3- minute step test) | 26 breast cancer patients receivinganaromatase inhibitor | Participantsreportedasignificantlylowernumberof painful joints, and improvedquality-of-life (p<0.05). Significant improvements in grip strength (p<0.01), bicepscurl (p<0.01), and sit-to-stand were also observed. Therewerenosignificantdifferencesinanthropometricmeasuresor cardiovascularendurance. |
Baruthetal., 2015 | 12-weekrandomizedcontrolledtrialofahome- based walking program | Self-reportedfatigue, quality- of-life, andwalking | 32 womenwhowereearly stagebreastcancersurvivors | Participantsintheinterventiongrouphadimprovementsinfatigueand qualityoflifeoutcomes. Changes in fatigue and quality of life were associatedwithchangesin walking behavior |
Total, leisure and vigorous physical activity significantly increased in the | ||||
Physicalactivity (primary | interventiongroupcomparedtousualcaregroup (p= 0.24, p=0.01, and | |||
Lahart et al., 2016 | 6-month randomized controlled trial of a physical activityintervention (face-to-face and telephone physical activity counseling) | outcome) andbodymass, bodymassindex, body fat , health-related quality-of-life, | 80 post-adjuvant therapy breastcancer patients | p=0.007, respectively). Both body mass and body mass index decreased significantlyintheintervention group compared to usual care group (p=0.04 and p=0.02, respectively). Total cholesterol and LDL-cholesterol |
insulinresistance, and lipids | decreasedsignificantlyinthe intervention group compared to usual care | |||
group (p=0.001 andp=0.23, respectively). | ||||
Knobfetal., 2016 | 12-monthrandomizedcontrolledtrial of a aerobic-resistance exercise intervention compared toa home-based physical activity intervention | Bonemineraldensity and biomarkersofboneturnover | 154 early postmenopausal breast cancer survivors | Nosignificant difference in bone mineral density was observed between the two groups |
Nyropetal., 2017 | 6-week randomized controlled trial of a home- based walking program | Self-reportedjointsymptoms and psychosocialmeasures | 62 post-menopausal women diagnosedwith stage 0-III breastcancer, with aromatase inhibitor-associatedarthralgia | Interventiongroupparticipantsreported significantly increased walking minutesperweek, reducedstiffness, less difficulty with activities of daily living, and less perceived helplessness in managing joint symptoms |
3-arm, 6-month randomized controlled trial of a | ||||
Valleetal., 2017 | physicalactivityintervention(activity tracker and tailored feedback based on objective weight; | Changeinweight | 35 AfricanAmericanbreast cancersurvivors, stageI-IIIa | Medianweightchangewas -0.9 intheintervention group that included activity trackers vs. 0.2% gain in the control group |
tailoredfeedbackalone; or control) | ||||
48-week multi-center randomized controlled trial | ||||
Westphaletal., 2018 | ofcounselingand unsupervised exercise training vs. supervised physical training (24 weeks) followedbyunsupervisedtraining (additional24 weeks). The supervised training was comprised of 45 minutes of stationary cycling and 30 | Maximumpower output on a cycleergometerafter 24 weeksofexercise | 42 early-stage breast cancer patientsreceiving aromatase inhibitortreatment | After 24 weeks, the supervised arm achieved a significantly higher maximumoutputinwatt (mean 132 +/- SD 34, 95% CI 117- 147) compared tobaseline 107 +/-25, 95% CI 97- 117, p=0.012) with a higher output than theunsupervised arm (115 +/- 25, 95% CI 105- 125, P=0.059). |
minutesofresistancetraining twice a week. | ||||
Randomizedcontrolledtrial of a home-based | ||||
Hirscheyetal., 2018 | physicalactivityintervention (a booklet about physicalactivityfor breast cancer survivors, that included narrative messages and writing and | Multidimensionalexercise outcomemeasure | 60 breast cancer survivors, stage Ia-IIb | Subjectiveexercise (weeklyminutes) increased 2 minutes, and objective exerciseincreasedby 970 steps, every 4 weeks in the intervention group comparedtothecontrolgroup (p=0.2676 and p=0.0283, respectively) |
thinkingexercises) | ||||
Vegetableconsumption, | ||||
Bailetal., 2018 | 12-monthrandomizedcontrolledtrialof a home- basedmentoredvegetablegardening intervention | physicalactivity, performance andfunction, anthropometrics, biomarkers, | 82 breastcancersurvivors, stage 0-III | Comparedwiththecontrols, interventionparticipants reported significantly greaterimprovementsinmoderatephysicalactivityanddemonstrated improvementsinthe 2-minutesteptestandarm curl (p-values<0.05). |
quality-of-life | ||||
Lahart et al., 2018 | 6-month randomized controlled trial of a home- basedphysical activity intervention | Cardiorespiratoryfitnessand physicalactivity | 32 breast cancer survivors whohadcompleted adjuvant therapy | Magnitude-basedinferenceanalysesrevealedatleastsmallbeneficialeffects onabsoluteandrelativeV̇O2 max (cardiorespiratoryfitness), andtotaland moderatephysicalactivityin the intervention compared to the usual care group. |
Yang etal.[16] conducted a 12-week randomized con-trolled trial of a home-based walking program among 40breastcancerpatientswho were receiving an aromatase inhibitor. Womenintheexercisegroupreportedsig- nificantly lower symptom severityscores (p<0.01) and mooddisturbance (p = 0.02) compared with those in thecontrolgroup.
Ina 12-week randomized controlled trial of a home- based, stage-matched, exercise and diet intervention (telephonecounselingandaworkbook), Kimetal.[17]foundthatthe intervention group showed significantly greaterimprovementinemotionalfunctioning(p = 0.04), andmotivationalreadinessforexercise (p<0.006) and dieting (p<0.001), and reduced fatigue (p = 0.001) and depression (p =0.035) than the controls.
In a 12-week randomized controlled trial of an email physicalactivityintervention, Hatchettetal.[18] ob-served significantlyhigherlevelsofphysicalactivityin the intervention group at six (p = 0.001) and 12 weeks (p<0.001) comparedto the controls.
Spectoret al.[19] conducted a pre/post-test trial of a home-basedaerobicandresistancetraining exercise in- tervention. Theinterventionincludedmotivationalinter- viewing and weekly telephone calls. Theyfoundasig- nificantincrease in total minutes of weekly physical ac- tivity (p = 0.001). Total quality-of-life and fatigue scores improved, but neither improvement was statistically sig- nificant.
Denysschen etal.[20] conducted an eight-week pre/post-testtrialofa home-based exercise program(resistanceexercises and self-selected aerobic exercise)among 26breast cancer patients who were receiving anaromataseinhibitor. The participants reported a signif- icantlylowernumberofpainfuljointsandsignificantly improvedquality-of-life(p<0.05). Significantimprove- mentsingripstrength (p<0.01), bicepscurl (p<0.01),and sit-to-stand were also observed. There were no sig-nificant differences in anthropometric measures or car-diovascularendurance.
In a 12-week randomizedcontrolledtrialofahome- based
walking program among 32 early-stage breast can- cer survivors, Baruth etal.[21] found that participantsin theinterventiongrouphadreducedfatigueandimprove- ment in other quality of life outcomes.
Lahartetal.[22] conductedasix-monthrandom- izedcontrolledtrialofaphysicalactivityintervention (face-to-face and telephone physical activity counseling)among 80 breast cancer survivors. Total, leisure, andvigorousphysicalactivityincreasedintheintervention group compared to the usual care group (p = 0.24, p = 0.01, and p = 0.007, respectively). Both body mass and bodymassindexdecreasedsignificantly in the interven- tiongroupcomparedtotheusualcare group (p = 0.04 and p = 0.02, respectively). In addition, total cholesterol and LDL-cholesterol decreased significantly in the inter- ventiongroup (p = 0.001) but not in the usual care group(p = 0.23).
Knobfetal.[23] conducted a 12-month randomized controlledtrialofanaerobic-resistanceexerciseinter- ventioncomparedtoahome-basedphysicalactivityin- terventionamong 154 early postmenopausal breast can- cer survivors. Theoutcomesofinterestwerebonemin- eral density and biomarkers ofboneturnover. Nosig- nificantdifferenceinbonemineral density was observed between the two groups.
Nyropet al.[24] conducted a six-week random- ized controlledtrialofahome-basedwalkingprogram among 62 post-menopausalbreastcancerpatientswith aromatase inhibitor-associatedarthralgia. Intervention groupparticipantsreportedsignificantlyincreasedwalk- ing minutes per week, reducedstiffness, lessdifficulty withactivitiesofdailyliving, andlessperceivedhelp- lessness in managing joint symptomscomparedtothe controls.
Valleetal.[25] conductedatwo-arm, six-monthran- domizedcontrolledtrialofaphysicalactivityinterven- tion (activity tracker and tailored feedback based on ob- jective weight; tailored feedback alone; orcontrol). The outcomeofinterestwaschangeinweight. Thirty-five AfricanAmericanbreastcancersurvivorsparticipatedin thetrial. Medianweightchangewas -0.9
% in the inter- ventiongroupthatincludedactivitytrackersvs. 0.2% gain in the controlgroup.
Westphaletal.[26]conducted a 48-week, multi-center,randomized controlled trialofcounselingandunsuper- visedexercisetrainingvs. supervisedphysicaltrain-ing (24 weeks) followedbyunsupervisedtraining (addi-tional 24weeks). The supervised training was comprisedof 45 minutes of stationary cycling and 30 minutes of re-sistancetrainingtwiceaweek. After 24 weeks, thesu-pervised arm achievedasignificantlyhighermaximumoutputinwatt (132 士 34, 95% CI:117- 147) comparedtobaseline 107 士 25, 95% CI: 97- 117, p= 0.012). Inaddition, output washigherinthesupervisedarm (115士25, 95% CI: 105- 125, p = 0.059) than in the unsuper-visedarm.
Hirscheyetal.[27] conductedarandomizedcontrolled trialofahome-basedphysicalactivityintervention (a bookletaboutphysicalactivityforbreastcancersur- vivors, thatincludednarrativemessagesand writing andthinking exercises) among 60 breast cancer survivors. Subjective exercise (weekly minutes) increased by two minutes, andobjectiveexercise increased by 970 steps every four weeksintheinterventiongroupcomparedto thecontrolgroup (p = 0.2676 andp = 0.0283, respec- tively).
Bailetal.[28] conducted a 12-month randomized con- trolledtrialofahome-basedmentoredvegetablegar-dening intervention among 82 breastcancersurvivors.Compared with the controls, intervention participants re-ported significantly greater improvementsinmoderate physicalactivityanddemonstratedimprovementsinthetwo-minutesteptestandarmcurl (p< 0.05).
Lahartetal.[29] conductedasix-monthrandom- izedcontrolledtrialofahome-basedphysicalactiv- ityinterventioninvolving 32 breastcancersurvivors. Magnitude-based inference analyses revealed at least smallbeneficialeffectsonabsoluteandrelativeVO˙2 max (cardiorespiratory fitness), andtotalandmoder- atephysicalactivityintheinterventioncomparedto the usualcare group.
4 Discussion
Theresultsofthissystematicliteraturereview indicate thathome-basedexerciseprograms are effective in im-
provingphysicalactivityamong breast cancer survivors whohavecompletedprimarytherapyfor the disease. A varietyofoutcomeswereassessedin the trials, including self-reportedminutesofphysicalactivity, stepswalked perday, functional capacity, cardiovascular endurance, bodymassindex, weight, sleep scores, quality-of-life, andattitudestowardphysicalactivity.In the one trial that assessedbonemineraldensity as an outcome[6] , no sig- nificant difference was observed between the two groups. Avarietyofinterventionstrategieshavebeentestedin trialsofhome-based exercise programs, including aero- bicandresistanceexercisetraining, walkingprograms, printmaterials,telephone counseling, and gardening. A majorityofthe outcomes measured in the trials using one ormoreoftheintervention strategies demonstrated posi- tivechangein the intervention groups compared to the controls. Thesestudiesyieldedencouraginginforma- tionandreflectedthe acceptance of innovative methods ofphysicalactivityinterventionsby breast cancer sur- vivors.
Threestudiesof the effectiveness of home-based exer- ciseprogramsfocusedon African American breast can- cersurvivors[11,19,25] . The results of these studies indi- catedthathome-based exercise programs are effective in increasingphysical activity and reducing weight amongAfricanAmericanbreastcancersurvivors.
Fourstudiesof the effectiveness of home-based exer- ciseprogramsfocused on women receiving aromatase inhibitortherapyforbreastcancer[20,24,26] . The results ofthesestudiesindicatedthathome-based exercise pro- gramsareeffective in reducing joint pain and increasing mobilityamongwomenreceiving this therapy.
TwoRCTs focused on using an email physical activity interventionamongbreastcancer survivors[17,18] . Both studieswereeffectiveusingemail,showing improve- mentwithmotivationreadiness for exercise, diet, emo- tionalfunctioning,depression and significant differences inphysicalactivity.
Fourstudiesonthe effectiveness of home-based walk- inginterventionsfocusedonbreast cancer survivors,
withoneofthe groups in each study receiving hormonal therapy[13–15,21] . These studies demonstrated that home- basedwalkinginterventionsare effective in increasing walking for exercise, reducing fatigue, and improving quality of life. Those receiving hormonal therapyre- portedimprovementinsleep scores and increased sero- toninlevels.
With respect to limitations, outcomes, intervention strategies, and tests varied among the studies. Caution isthereforerequiredin comparing results across studies. Inaddition, our literature review may not have captured allrelevantstudies.
Insummary, home-based exercise programs are effec- tivein improving physical activity among breast cancer survivorswho have completed primary therapy for the disease. This includes women who are receiving aro- mataseinhibitortherapyforbreast cancer. Home-based exercise programs, such as walking programs offera convenientandaffordableoptionforwomen who wish toincreasetheirphysicalactivityandmaintaina healthy lifestyle.
Conflictofinterest
The authors declare they have no conflicts of interest.
Ethicalapproval
Thisarticledoesnotcontainany studies with human participantsperformed by any of the authors.
References
[1] American Cancer Society. Cancer Facts & Figures 2019. Atlanta:American Cancer Society, 2019.
[2] Zhang X, Li Y and Liu D. Effects of exercise on the qualityoflifeinbreastcancerpatients: a systematic review of ran-domizedcontrolledtrials. Supportive Care in Cancer, 2019,27(1): 9-21.https://doi.org/10.1007/s00520-018-4363-2
[3] World Cancer Research Fund Report “Food, Nutrition,Physical Activity, and the Prevention of Cancer: a Global
Perspective” .2007, 1(10): 464-469.https://doi.org/10.1007/s12082-007-0105-4
[4] VanceV, MourtzakisM, McCargarL, etal. Weightgainin breast cancer survivors: prevalence, pattern and healthconsequences. Obesity Reviews, 2011, 12(4): 282-94.https://doi.org/10.1111/j.1467-789X.2010.00805.x
[5] Rock CLandDemark-WahnefriedW. NutritionandSur-vivalaftertheDiagnosisof
BreastCancer: AReviewoftheEvidence. Journalof Clinical Oncology, 2002, 20(15):3302-3316.https://doi.org/10.1200/JCO.2002.03.008
[6] KnobfMT, ThompsonSA, FennieK, etal. The Effect of aCommunity-Based Exercise Intervention on Symptoms and
Quality of Life. Cancer Nursing, 2014, 37(2): E43-E50.https://doi.org/10.1097/NCC.0b013e318288d40e
[7] SmithSA, AnsaBE, Yoo W, etal. Determinants of ad- herencetophysicalactivityguidelinesamongoverweightandobeseAfricanAmerican breast cancer survivors: im-pliationsfor an intervention approach. Ethnicity & Health,2018, 23(2): 194-206.https://doi.org/10.1080/13557858.2016.1256376
[8] Coughlin SS and Smith SA. The insulin-like growth factoraxisadipokines, physicalactivity, andobesityin relation tobreastcancer incidence and recurrence. Cancer and ClinicalOncology, 2015, 4(2): 24-31.https://doi.org/10.5539/cco.v4n2p24
[9] Schmidt T, van Mackelenbergh M, Wesche D, etal. Physicalactivity influences the immune system of breast cancer pa-tients. Journalof Cancer Research and Therapeutics, 2017,13(3): 392-398.https://doi.org/10.4103/0973-1482.150356
[10] PintoBM, Frierson M, Rabin C, etal. Home-based physicalactivityinterventionforbreastcancerpatients. JournalofClinicalOncology,2005, 23(15): 3577-3587.https://doi.org/10.1200/JCO.2005.03.080
[11] WilsonDB, PorterJS, ParkerG, etal. Anthropometricchanges using a walking intervention in African American breastcancersurvivors: a pilot study. Preventing ChronicDisease, 2005, 2(2): 1-7.https://www.cdc.gov/pcd/issues/2005/apr/040112.htm
[12] VallanceKH, Courneya KS , Plotnikoff RC, etal. Random-ized controlled trial oftheeffectsofprintmaterialsand steppedometersonphysicalactivityandqualityoflife inbreastcancersurvivors. JournalofClinical Oncology, 2007,25(17): 2352-2359.https://doi.org/10.1200/JCO.2006.07.9988
[13] MatthewsCE,Wilcox S, Hanby CL, etal. Evaluation of a12-weekhome-based walking intervention for breast cancersurvivors. Supportive Care in Cancer, 2007, 15(2): 203-211.https://doi.org/10.1007/s00520-006-0122-x
[14] Yuen HK and Sword D. Home-based exercise to alleviate fa- tigueandimprovefunctionalcapacity among breast cancer survivors. Journal of allied health, 2007, 36(4): e257-275.
[15] Payne JK, Held J, Thorpe J, etal. Effect of exercise onbiomarkers,fatigue, sleep disturbances, and depressivesymptomsin older women with breast cancer receiving hor-monaltherapy. OncologyNursingForum, 2008, 35(4): 635-642.https://doi.org/10.1188/08.ONF.635-642
[16] Yang CY, Tsai JC, Huang YC, etal. Effects of a home-based walkingprogramonperceivedsymptomand mood status inpostoperativebreastcancerwomenreceivingadjuvant chemotherapy. Journal of Advanced Nursing, 2011, 67(1):158-168.
https://doi.org/10.1111/j.1365-2648.2010.05492.x
[17] Kim SH, Shin MS, Lee HS, etal. Randomized pilot test ofasimultaneousstage-matchedexercise and diet interventionforbreastcancersurvivors. Oncology Nursing Forum, 2011,38(2): E97-E106.https://doi.org/10.1188/11.ONF.E97-E106
[18] Hatchett A, Hallam JS and Ford MA. Evaluation of a so-cialcognitive theory-based email intervention designed toinfluencethephysicalactivityofsurvivors of breast cancer.Psycho-Oncology, 2013, 22(4): 829-836.https://doi.org/10.1002/pon.3082
[19] SpectorD, DealAM, AmosKD, etal. Apilotstudyof a home-basedmotivationalexerciseprogramforAfricanAmericanbreastcancersurvivors: clinicalandquality-of-lifeoutcomes. IntegrativeCancerTherapies, 2014, 13(2):121-132.https://doi.org/10.1177/1534735413503546
[20] DenysschenCA, BurtonH, AdemuyiwaF,etal. Exer- ciseinterventioninbreast cancer patients with aromataseinhibitor-associatedarthralgia: apilotstudy. European Jour-nalofCancerCare,2014, 23(4): 493-501.https://doi.org/10.1111/ecc.12155
[21] BaruthM, WilcoxS, DerAnanianC, etal. Effectsof home-basedwalkingonqualityoflifeandfatigueout- comes inearly stage breast cancer survivors a 12-week pilot study. Journalof Physical Activity and Health, 2015, 12(s1):
S110-S118.https://doi.org/10.1123/jpah.2012-0339
[22] Lahart IM, Metsios GS, Nevill AM, etal. Randomized co
n-trolled trial of a home-based physical activity intervention inbreast cancer survivors. BMC Cancer, 2016, 16: 234.https://doi.org/10.1186/s12885-016-2258-5
[23]KnobfMT, JeonS, SmithB, etal. Effectofarandom-izedcontrolledexercise trial on bone outcomes: influenceof adjuvant endocrine therapy. Breast Cancer Research andTreatment, 2016, 155(3): 491-500.https://doi.org/10.1007/s10549-016-3693-3
[24] NyropKA, Callahan LF, Cleveland RJ, etal. Randomized controlledtrial of a home-based walking program to reduce moderatetoseverearomataseinhibitor-associated arthralgia inbreast cancer survivors. The Oncologist, 2017, 22(10):
1238-1248.https://doi.org/10.1634/theoncologist.2017-0174
[25] Valle CG, Deal AM and Tate DF. Preventing weight gain inAfricanAmericanbreast cancer survivors using smart scalesandactivity trackers: a randomized controlled pilot study.JournalofCancer Survivorship, 2017, 11(1): 133-148.https://doi.org/10.1007/s11764-016-0571-2
[26] Westphal T, Rinnerthaler G, Gampenrieder SP, etal. Su-pervisedversusautonomousexercisetraininginbreast can-cerpatients:a multicenter randomized clinical trial. CancerMedicine, 2018, 7(12): 5962-5972.https://doi.org/10.1002/cam4.1851
[27] Hirschey R, Kimmick G, Hockenberry M, etal. A randomizedphaseIItrialofMOVINGON: Aninterventionto in-creaseexerciseoutcomeexpectations among breast cancersurvivors. Psycho-Oncology, 2018, 27(10): 2450-2457.https://doi.org/10.1002/pon.4849
[28] BailJR,Fruge AD, Cases MG, etal. A home-based men-toredvegetablegardeningintervention demonstrates feasibility and improvements in physicalactivityandper-formance among breast cancer survivors. Cancer, 2018,124(16): 3427-3435.https://doi.org/10.1002/cncr.31559
[29] Lahart IM, Carmichael AR, Nevill AM, etal. The effects of ahome-basedphysicalactivityinterventionon cardiorespi- ratory fitnessinbreast cancer survivors; a randomised con- trolledtrial.Journal of Sports Sciences, 2018, 36(10): 1077-
1086.https://doi.org/10.1080/02640414.2017.1356025